Deterioration of Concrete Its Causes and Prevention | Leaching Action |Chemical Reaction | Crystallization | Corrosion of Reinforcement
Hello Guys today we will see what deterioration of concrete and its causes and how to prevent them in detail. I hope you like this article.
What is deterioration of concrete ?
Deterioration of concrete means due to various factor like leaching action , chemical reaction , crystallization and corrosion of concrete reduce the durability of concrete which effect overall strength of structure.

Deterioration of concrete:
There are two main reasons for low durability of concrete.
1) Deterioration Of Concrete
2) Corrosion Of Reinforcement
These two phenomena cannot be separated from each other, as the corrosion of reinforcement is due to the deterioration of the concrete cover. On the other hand, corrosion of reinforcement (rods) causes internal stresses in the surrounding concrete and further breaks the concrete around the rods.
Concrete has very fine cracks. If the number of these microscopic cracks is low. And breakage does not affect the durability of concrete, but when these cracks become continuous and wide due to internal factors, aggressive ions and gases easily reach the reinforcement, corrode it and reduce the durability of concrete.
Types of Deterioration:
Concrete is degraded not only by acids or acidic gases in the form of water solution, but also by salt solutions and alkalis. Other elements such as chemical fertilizers, pesticides, and some organic compounds are also harmful to concrete.
Concrete deterioration caused by corrosion of rods by different aggressive chemicals can be divided into three groups.
There are three main forms of concrete destruction.
(i) Leaching Action
(ii) Chemical Reaction
(iii) Crystallization
(i) Leaching Action:
This type of destruction is caused by dissolving frozen cement components in aqueous solution. The calcium hydroxide of hard cement is soluble, hence the destruction of concrete by leaching action, also known as lime leaching. Lime leaching depends on the permeability of the concrete. When the free lime in the concrete is leached, the hydrolysis of calcium silicate and aluminate releases more lime which is re-leached.
 |
|
Leaching Action |
The presence of salt in the solution affects the dissolution rate of calcium hydroxide. For example, the presence of the same ions (Ca++),( OH-) slows down this rate, while other ions such as (SO-24),(CL– ), (Na+), (K+) etc. accelerate this rate.
 |
| Leaching Action |
Leaching lime react with carbon dioxide and Forms calcium carbonate, so white calcium carbonate is spread on the surface of the concrete, which is called the death of concrete from leaching.
(ii) Chemical Reaction:
Calcium salt is formed by the chemical reaction between hardened cement constituents and acid solution or salt solution. The product of a chemical process is removed from the concrete by diffusion process or percolation if soluble in water. But if the product is not water soluble it sticks to the concrete surface which can be easily washed.
 |
| Chemical Reaction |
 |
| Deterioration of concrete due to acid attack |
It is clear from Figure above that low doses of hydrochloric acid (HCI) do more to break down concrete than sulphuric acid (H2SO4). This is because the calcium salt (CaSO4) produced by the chemical reaction of H2SO4 with the lime of the concrete is less soluble, which fills the cavities of the concrete and prevents the breakdown of the concrete sue to acid.
(iii) Crystallization:
Crystallization occurs when salt accumulatesin the cavities of concrete. This causesinternal stresses in the concrete and cracks in the concrete. These salts that accumulate in concrete cavities are either produced by chemical process or enter the cavity by salt solution from outside. Concrete breaks down quickly due to simultaneous drying and wetting of the concrete memberscontaining the soluble solution. Magnesium ions, together with sulphate ions (MgSO4) break down concrete faster, as crystallization of gypsum increases the permeability of concrete.
 |
|
Crystallization |
Prevention of concrete Deterioration:
The following two things are very important for the durability of concrete.
(1) Proper bonding agent which has good resistance to chemicals.
(2) Adequate compaction of concrete to get its high density
(3) Information on water / cement ratio, cement ratio, curing conditions, use of admixtures, etc. is useful to prevent concrete deterioration.
(4) If water cement ratio increase from 0.45 to 0.5 then it increase permeability too high Therefore the water-cement ratio should be less than 0.45 – 0.55.
(5) The amount of cement for a given water / cement ratio depends on the need for workability . Also, the amount of cement should be such that the required pH value of the concrete is obtained and the reinforcement passive atmosphere occur against erosion.
(6) The minimum cement content in concrete used in seawater or coastal environments should be 350 kg/m.
(7) In addition, the water / cement ratio and cement ratio should be such that the cement paste produced can fill the cavities of the aggregate, the proportion of cavities or voids in the aggregate depends on the type of aggregate and the maximum normal size. E.g. , 20 mm size aggregate of crushed rock has 27% void ratio and 20 mm size aggregate of river round gravel has 22% void ratio.
(8) From the use of 0.45 water / cement ratio and 400 kg/m3 The size of the paste produced from cement is about 30%, which is enough to fill the cavities of the aggregate of broken stone.
(9) On the other hand, the paste produced from 0.50 water / cement ratio and 300 kg / m cement has a size of 25%. Which is enough to fill the cavities of the circular gravel of the river.
(10) At least M – 15 concrete grade for PCCand at least M -20 concrete grade for RCC should be used in seawater or coastal concrete.
(11) Such concrete should use Portland slag-cement or Portland Pozzolona cement. Super sulphate cement is also beneficial.
(12) Deterioration of concrete can be prevented by creating a natural or artificial carbonated layer on the surface. The carbonated layer prevents the deterioration of concrete by leaching.
(13) Applying a solution or a small amount of acid to the surface of the concrete produces a layer of calcium salt which is less soluble than calcium hydroxide so that the deterioration of the concrete stops.
(14) The durability of concrete can also be enhanced by inserting a polymer into the cavity.
Corrosion of Reinforcement:
Corrosion of reinforcement is a very complex process. Which includes chemical, electrochemical and physical processes. When reinforced steel is corrugated, iron oxide is produced which is 2 to 4 times the size of the corrugated steel.
 |
Corrosion of Reinforcement |
Thus in concrete, high pressure are produced around the rod. So that the concrete cracks and breaks. Secondly, corrosion reduces the cross section of the rod and also reduces its ability to withstand tensile forces.
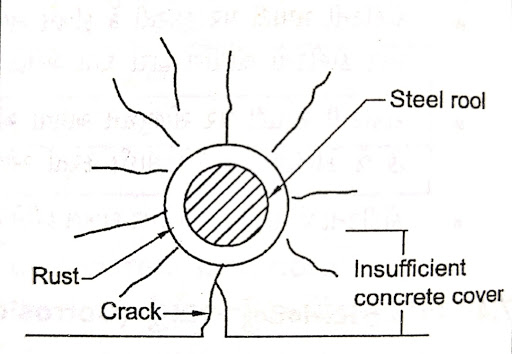 |
| Cracking due to rust |
To corrosion of the rod must first satisfied an electrochemical cell . This cell has two electrodes – anode and cathode. These two electrodes are separated by electrolytes and connected in an electric circuit.
On the surface of reinforcement steel, the part where the most oxygen is concentrated becomes the cathode and the part where the less oxygen is concentrated becomes the anode.
 |
||
|
Concrete always has a small amount of moisture which acts as an electrolyte. Concentration of salt in water in cavities in concrete also forms electrochemical cells. The part with the highest concentration of salt becomes the anode, while the part with the least concentration of salt becomes the cathode.
This is the potential difference between anode and cathode. Which, in the presence of water and oxygen, oxidizes the steel to form iron oxide. The + charged ferrous ions near the anode move into the (Fe ++) solution while the charged free electron (e) passes through the steel and reaches the cathode.
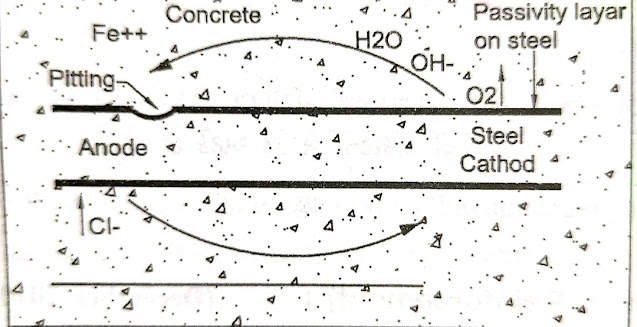 |
| Corrosion mechanism and electrochemical cell |
These free electrons (e) form hydroxyl ions (OH) together with water and oxygen near the cathode. This (OH) ion combines with (Fe ++) ions to form ferrous oxide Fe (OH)2. Re-oxidation of ferrous oxide results in the formation of ferric oxide Fe (OH)3 followed by corrosion.
Causes of Corrosion and Preventive measures):
The main causes of steel corrosion and prevention measures are as under:
1) 1) Cracks in concrete
2) 2) Presence of moisture
3) 3) Permeability of concrete
4) 4) Carbonation
5) 5) Chlorides
6) 6 )Sulphate attack
7) 7) Alkali – Aggregate reaction
8) 8) Inadequacy of cover
1) 1) Cracks in concrete:
Oxygen and seawater enter through cracks in concrete and create a suitable environment for corrosion. For normal environments, the width of the cracks in the protected inside members should not exceed 0.30 mm and the width of the cracks in the unsafe outside members should not exceed 0.20 mm.
2) Presence of Moisture:
The presence of moisture in concrete is essential for the corrosion of steel, as concrete can act as an electrolyte in an electrochemical cell only if there is moisture. Corrosion cannot occur in completely dry or submerged concrete. Therefore low permeability concrete should be used to prevent corrosion.
3) 3) Permeability of concrete:
If the concrete is porous, moisture, oxygen, CO2, seawater can easily enter it. If the water / cement ratio increases by 0.1, the permeability of concrete increases 1.5 times.
Inadequate curing also increases the permeability of concrete by 5 to 10 times.
Inadequate pressure also increases the permeability of concrete by 7 to 10.
To reduce the permeability of concrete, minimum cement ratio should be 350 kg / m and maximum water / cement ratio should be 0.45. In addition, adequate attention should be paid to aggregate grading, mixing , compaction curing.
4) Carbonation:
Carbon dioxide (CO2) in the air enters the concrete and combines with calcium hydroxide to form calcium carbonate, which is soluble in water. This process is called carbonation.
Carbonation reduces the alkalinity of concrete and its resistance to corrosion. The pH value of water in concrete cavities is 10.5 to 12.0. But with carbonation it dropped to 9.0. Is reduced, thus creating a favourable environment for corrosion.
Carbonation in concrete penetrates to a depth of 100 mm in 50 years. To prevent corrosion of concrete by carbonation, it is necessary to cover the steel with at least 20 mm. Good quality, high density concrete should be used.
5) 5) Chlorides:Chlorides enter concrete during concreting or during use.
Chloride content in aggregate for RCC should not exceed 0.05% and calcium chloride content in mixing water should not exceed 1000 mg / Lit (PPM).
The concentration of Calcium Chloride (CaCI2) used as admixtures in concrete should not exceed 1.5%.
In addition, adequate attention should be paid to steel adequate cover, good quality concrete, aggregate with good grading.
6) Sulphate attack:
Soluble sulphates like sodium, potassium, magnesium, calcium etc. are present in water, soil or bricks.
The volume when these sulphates combined with C3A and lime of cement is very large.
So that the concrete cracks and the steel corrodes.
For barrier against sulphate attack,
*concrete should not be less than M – 20 grade.
*Sulphate resisting cement (CA less than 3.5%) should be used.
*Cement content should not be less than 350 kg / m³ and water / cement ratio should not be more than 0.45
7) Alkali aggregate reaction:
OPC contains alkalis like sodium oxide (Na2O) and potassium oxide (K20). These alkalis process with some reactive aggregates (traps, andesites, phyolites, silicious limestone, dolomite) called alkali aggregate reaction.
This process causes the concrete to expand and crack. To prevent the effect of an alkali aggregate reaction . Reactive type aggregates should not be used.
(8) Insufficient concrete cover:
If the concrete cover is insufficient, the steel will be corroded by sea water, moisture, carbonation. The following cover is recommended in concrete as per IS 456-2000.
1)Should not be less than the diameter of the rod.
2) The column should not be less than 40 mmfor the main rod.
3)Under no circumstances should it be less than 20 mm.
4) Must be no less than 50 for footing.
In addition, the following steps are taken to prevent corrosion of steel.
(i) Application of Protective coating
(ii) Modification of concrete
(iii) Change in Metallurgy of steel
(iv)Cathodic Protective stystem.
+ READ MORE




